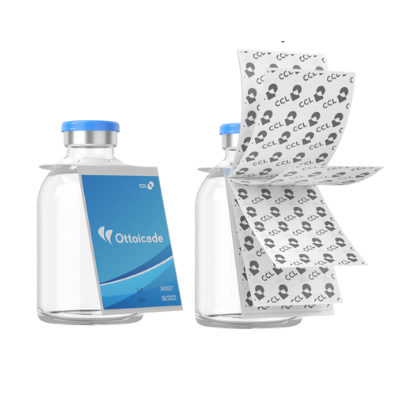
Serialized Topsert Leaflets - Overview
Printed Literature

Printed Literature - Serialized Topserts
Serialized Topsert leaflets are a type of folded packaging insert that is placed on top of the product within its packaging. It typically contains important information such as dosage instructions, side effects, and precautions, and is an essential component of the packaging for many pharmaceutical and healthcare products.
Serialization is a process of assigning a unique code or serial number to each individual product or package. This code is used to track and trace the product throughout its lifecycle, from manufacturing to distribution to the end user. Serialization is an important tool for ensuring product safety and integrity, and is becoming increasingly important in the pharmaceutical and healthcare industries.
By combining a topsert leaflet with serialization, pharmaceutical manufacturers can provide their customers with an additional layer of security and assurance. Each individual leaflet can be assigned a unique serial number that corresponds to the product it accompanies, allowing for easy tracking and tracing of both the product and the leaflet itself.
This can be particularly useful in cases where a product recall is necessary, as it allows the manufacturer to quickly identify which products are affected and take appropriate action. Serialization can also help to prevent counterfeiting and protect against theft or diversion of the product.
Folded Outserts for Patient Information
Patient information leaflets, also known as PILs, are an essential component of any medication. They provide critical information about the medication, its benefits, possible side effects, dosage instructions, and precautions. However, sometimes the information is too lengthy to fit onto a single sheet of paper. In such cases, pharmaceutical companies use folded leaflet outserts, which contain more extensive information than a standard leaflet. In this blog, we’ll discuss patient information folded leaflet outserts and their significance.
A folded leaflet outsert is a booklet-style document that is often inserted inside a medicine packet. These leaflets provide detailed information about the medicine and are essential for patients to understand the medication’s effects, side effects, dosage, and administration instructions. Unlike a standard leaflet, which typically consists of two to four pages, folded leaflet outserts can contain up to 32 pages, providing ample space to include all the necessary information.
These leaflets are generally made from a single sheet of paper, which is folded multiple times to create several panels, with each panel containing vital information about the medication. Typically, the front panel of the leaflet contains the brand name of the medication, its active ingredients, and the dosage. The following panels provide information about how to take the medication, potential side effects, and warnings and precautions. The last panel often includes details about the manufacturer and how to report adverse effects.
Contact Us
Contact a Packaging Specialist.
Request a Quote
Do you need a quote for Printed Literature?
Newsletter
Stay up-to-date with CCL Healthcare.
Printed Literature Overview
Printed Literature from CCL is printed in an array of options to meet regulatory, multilingual, and instructional demands in the healthcare packaging industry. Choose from a wide range of high-capacity folded package inserts, outserts and padded inserts to fit your needs. IFU / DFU booklets can be glued or stitched in a variety of sizes.
- Inserts IFU / DFU
A multi-folded leaflet formed from a single sheet of paper, for insertion into cartons providing space for detailed legislative and patient information with scope for multiple languages.
- Outserts IFU / DFU
A married outsert (twinsert/piggyback) is created by gluing one or more outserts together to create a bundle. The outserts can be a combination of physician and patient information and allows for multiple copies or additional package information.
- Glued Pads
Flat or multi-folded single sheets of paper are combined and glued along one edge to create a pad, which can be inserted into cartons or applied to neck hangers.
- Booklets
Booklets are produced with a variety of stocks, colors and page counts. Booklets are an effective way to distribute large amounts of regulatory information.