Cryogenic Labels
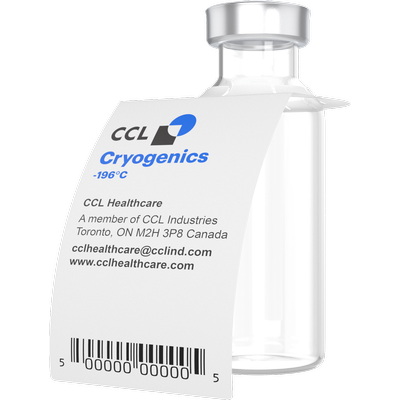
Cryogenic Slit Label

Cryogenic Slit Label on a Cryo Vial
Cryogenic slit label on a cryo vial is hand feed through the die cut slit. The leading edge adheres back onto the cryogenic slit label securing the label. The slit on the label adds more tension to the cryo vial.
Every cryogenic slit label is made with durable materials, adhesives, and ink adherence to withstand the extreme temperature excursions during its life cycle. Cryogenic slit labels can be customized based on your product’s specific needs including: constructions, materials, imprinting method, and color combinations, delivering the most effective solution for your product requirements.
Overview of Cryogenics
Contact Us
Contact a Packaging Specialist.
Request a Quote
Do you need a quote for Cryogenic Labels?
Newsletter
Stay connected with CCL Healthcare.
Why CCL for Cryogenics?
CCL Healthcare offers a vast selection of cryogenic product solutions along with a strategy that can boost your sales and add value to your product. Our product line grows as the demand for innovative solutions increases. Our pharmaceutical and healthcare cryogenic products are printed with the most advanced equipment available in the industry. Pairing our highly trained and qualified staff with our robust SOP’s and cGMP facilities allows us to exceed our customers’ quality expectations.
Our investment in equipment and people has allowed us to continually be the market innovators and grow our labeling, printed literature, and carton offerings to meet the unique challenges that the pharmaceutical and healthcare industry demands. Our product lines are custom-designed, specifically to customers’ formats and requests. The product line includes a variety of cryogenic products such as cryogenic booklet labels on vials, cryogenic flag labels, cryogenic neck hanger labels, cryogenic overlap labels on glass vials, cryogenic slit labels on cryo vials, cryogenic wraparound labels on cryo vials, and other custom products.
When it comes to quality CCL Healthcare is the market leader investing heavily into vision verification systems and establishing the standard for pharmaceutical printing in the industry. The standard includes ensuring every product is 100% verified and documented for traceability. We manufacture under cGMP and ISO standards in temperature and humidity control facilities. Our dedicated quality assurance team and production departments follow strict SOPs and CSPs in fully segregated manufacturing areas. Our global presence and manufacturing footprint allows you to go global with 34+ specialized healthcare sites. Our experienced staff provides the highest level of service, technical expertise, and product innovation anywhere you are.