Clinical Labeling
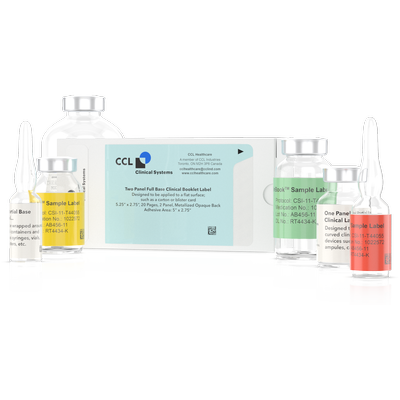
16 Page Booklet Label (ECL)

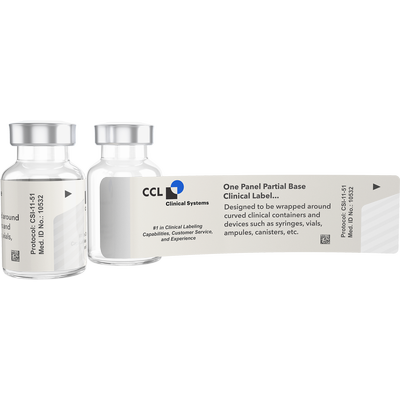
One Panel Full Base 16 Page Booklet Label
The 16-page Clinical Booklet Label with One Panel, Full Base is designed to be applied to flat surfaces, such as cartons, blister cards, and kits. Full base labels can be created with a clear base or opaque base. During clinical trials clear bases and opaque bases are utilized in the distribution of medication. This 16-page clinical booklet label, with one panel full base is designed using a clear base for applications when masking is not necessary.
About Clinical Booklets
Clinical Booklet Labels are used when conducting global clinical trials where flexibility to distribute the clinical supplies to a range of international locations is necessary. This is accomplished by creating a booklet of pages that contain label text for multiple languages, positioned inside a reseal-able cover. The clinical booklet labels can then be adhered to various packages for multinational trials. The labels are designed in several different configurations dependent upon the clinical dispenser or layer of packaging they are being applied to. All our booklet labels can be opened and resealed to gain access to the data on the individual pages when required.
Browse Similar Clinical Booklet Labels
Quality Built Into Every Product & Process
CCL Healthcare adheres to strict guidelines in all of our manufacturing processes and conforms to Good Manufacturing Practices (GMP) to achieve manufacturing excellence and deliver the highest quality products for every client, every time. We’ve established effective processes that are clearly defined and controlled to ensure consistency and compliance with all your job specifications. Our quality systems have been integrated into every step of our process to assure that each critical interaction is controlled with precision. SOP’s are continually reviewed and updated as necessary and all of our staff receive extensive SOP training throughout each year.
When you contract with CCL Healthcare, you can be assured that quality is built into every finished product and into every step of the manufacturing process as well.
Learn More About Clinical Booklet Labels
Bases are available in white, opaque or clear. White label bases are used for labeling new blank components. Opaque base labels can be used to cover up existing materials or other data such as pre-stamped lot information. Clear base labels are used when it is necessary to see into the container to use the product. The picture of the vial is of a clear base label.
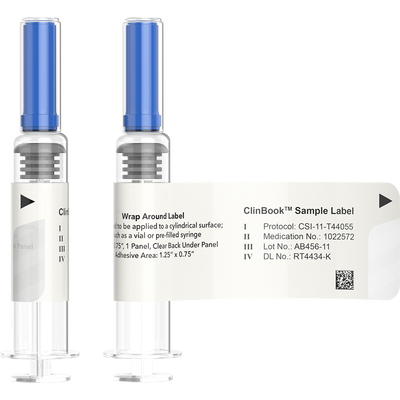
Multiple removable panels with adhesive backing can also be incorporated into all of the booklet label designs. These panels can be used to verify drug usage in case books or document other clinical requirements at the time of distribution or dispensing. Many clinical trials necessitate the need for cold temperature storage of the drug entities. We can manufacturer these labels with cryo adhesives that are specifically designed for these types of environments.
Clinical Booklet labels can be provided on rolls for automatic or hand application, or fanfolded. In each case, we label the roll or bundle with starting and ending variable data labels so that you know which label segment is contained on each unit of supply. Covers of booklets can be supplied with full cover tints to provide clinical differentiation of similar materials. Variable data can be printed on each label including randomization codes, lot information and dating as text or and as barcodes. 2-D and linear barcode symbologies can be implemented.